sCD25 and Ferritin Levels Best Distinguish Children with Hemophagocytic Lymphohistiocytosis from Relevant Controls
Bethany Verkamp, MD; Adi Zoref-Lorenz, MD; Dafna Brik Simon, MD; Joanne Yacobovich, MD; Joana Mack, MD; Tucker Blackledge, MD; Michael Jordan, MD
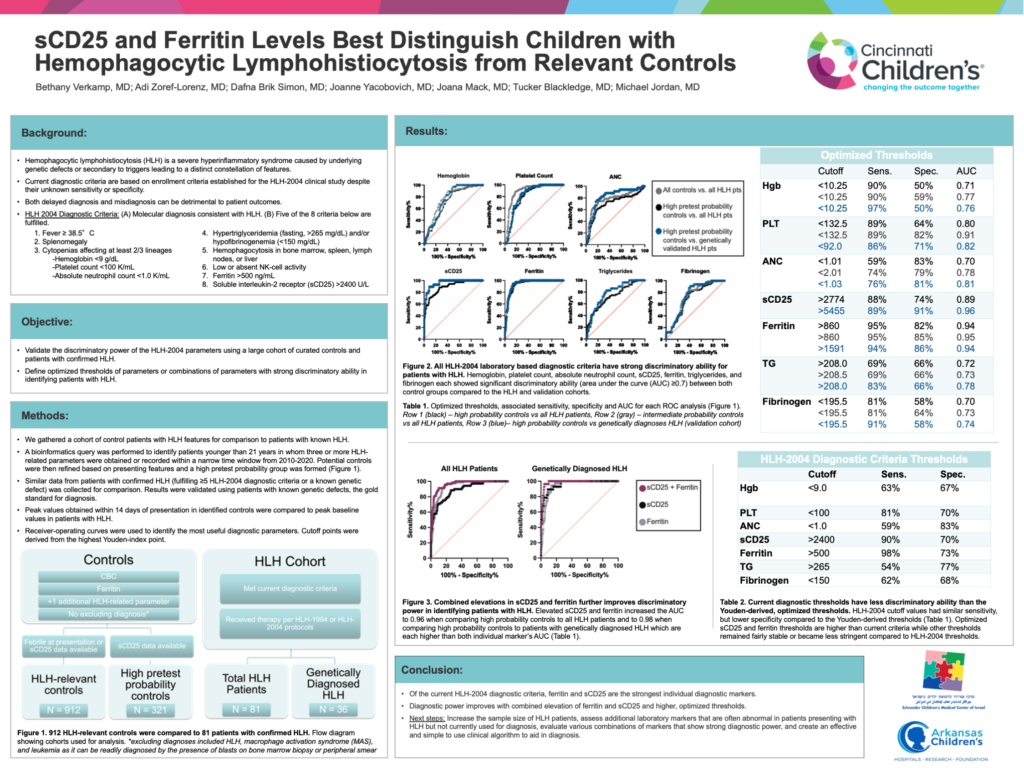
sCD25 and ferritin are the two strongest diagnostic markers for identifying patients with HLH. A combined elevation of these two markers and the use of higher threshold values further improves their discriminatory ability.
Bethany Verkamp, MD
Abstract
Background: Hemophagocytic lymphohistiocytosis (HLH) is a severe hyperinflammatory syndrome caused by underlying genetic defects or secondary to triggers leading to a distinct constellation of features. Current diagnostic criteria are based on enrollment criteria established for the HLH-2004 clinical study despite their unknown sensitivity or specificity.
Objective: Validate the discriminatory power of the HLH-2004 parameters using a large cohort of curated controls and patients with confirmed HLH.
Methods: We gathered a cohort of control patients with HLH features for comparison to patients with confirmed HLH (fulfilling ≥5 HLH-2004 diagnostic criteria or a known genetic defect). Controls included patients younger than 21 years in whom a CBC, ferritin, and one additional HLH-related parameter was obtained within a narrow time window from 2010-2020. Patients presenting with fever were defined as intermediate pretest probability controls. Patients who had soluble CD25 (sCD25) levels examined were defined as high pretest probability controls. Peak values obtained within 14 days of presentation were compared to peak baseline values in patients with HLH. Receiver-operating curves were used to identify the most useful diagnostic parameters. Cutoff points were derived from the highest Youden-index point. Results were validated using patients with known genetic defects, the gold standard for diagnosis.
Results: We identified 18,204 potential controls, 907 intermediate pretest probability controls, and 321 high pretest probability controls. Hemoglobin, platelet count, absolute neutrophil count, sCD25, ferritin, triglycerides, and fibrinogen showed significant discriminatory ability (area under the curve (AUC) ≥0.7) between the HLH cohort and both control groups. The individual parameters with the greatest discriminatory power were sCD25 (AUC 0.89) and ferritin (AUC 0.95 compared to intermediate pretest probability controls, AUC 0.94 compared to high pretest probability controls). Discriminatory ability further improved with a combined elevation of sCD25 and ferritin (AUC 0.96). Optimized thresholds of sCD25 and ferritin were higher than HLH-2004 cutoffs in each analysis. Moreover, when comparing controls to genetically diagnosed patients, sCD25 had the highest discriminatory power with an AUC of 0.96 and with ferritin, a combined AUC of 0.98.
Conclusion: Of the HLH-2004 criteria, sCD25 and ferritin are the strongest individual diagnostic markers. Diagnostic power further improves with combined elevation and higher threshold values.